 
the maximum possible amount of water vapor in the air), but the partial pressure of the water vapor is unchanged. Because of this heating, the saturation pressure of the water vapor rises (i.e. As this air enters a building it is heated to +21 ☌, but the amount of water remains constant – no water is removed or added to the air in normal ventilation systems. Let’s imagine that the outside temperature on a crisp winter day is -14 ☌, and the relative humidity is 60%. This, of course, means that relative humidity is also strongly temperature dependent. In other words, relative humidity always lies between 0 and 100%.Īs mentioned above, the ability of air to hold water vapor is strongly dependent on temperature. On the other end of the scale, when there is no water vapor in the air, the relative humidity is 0% whatever the temperature. At this point, the air is said to be saturated with water, and the relative humidity is 100%. If the maximum amount of water vapor has been reached and more water is introduced into the air, an equal amount of water must transform back to liquid or solid form through condensation. 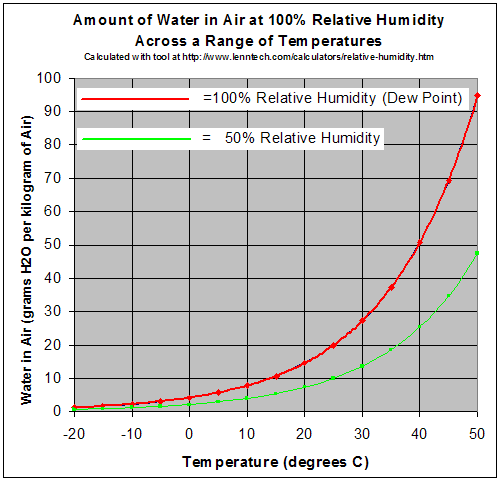
In other words, relative humidity, expressed in per cent, is the partial water vapor pressure in relation to the saturation pressure. The ‘relative’ in relative humidity expresses the relation between the amount of water vapor present and the maximum amount that is physically possible at that temperature. ‘Relatively’ few people, however, understand what it really means. Relative humidity is the most commonly used humidity unit. The atmospheric pressure at an altitude of 8,800 meters is only about one-third the sea level pressure, so the tea water would boil at well below 70 ☌. If the mountain climber made himself a cup of tea on top of Mount Everest, the taste would probably leave something to be desired. In this light, the boiling point of a liquid is dependent not only on the physical properties of the liquid, but also on the surrounding atmospheric pressure. Water Vapor Saturation Pressure CurveĪt 100 ☌, the boiling point of water, the water vapor pressure surpasses normal atmospheric pressure. The partial water vapor pressure in the immediate presence of liquid water equals the saturation pressure at that specific temperature.įigure 1. The higher the temperature, the higher the partial pressure of the water vapor. The ability of water to be in gaseous form is strongly dependent on its temperature (see Figure 1: Water vapor saturation pressure curve). The air temperature dictates the maximum partial water vapor pressure in air, in other words, the water vapor saturation pressure. On this basis, 21% of the total volume of dry air is oxygen and around 1% is typically argon. All gases produce the same pressure and volume with the same number of molecules, so the partial pressures also represent the proportion by volume of the various gases. The atmospheric pressure, usually around 1000 hPa, is the total of the partial gas pressure of nitrogen (~775 hPa), oxygen (~205 hPa), water vapor (~10 hPa), argon (~10 hPa) carbon dioxide (~0.4 hPa) and a number of other gases with lower partial pressures. In a gas mixture such as air, the total pressure (same as atmospheric or barometric pressure) of the gas is the sum of all the individual pressures of its gas components. Properties of Gas MixturesĪ full understanding of the various terms for humidity and moisture requires some basic knowledge about the properties of gas mixtures. The term moisture also applies to extremely dry gases, when water vapor is considered an impurity. 
Moisture, on the other hand, is used for liquids and solid materials. When the term humidity is used, we usually mean water vapor in a gas, typically air. Partial water vapor pressure, absolute humidity, frostpoint, mixing ratio, wet bulb temperature and even enthalpy all describe the humidity of a gas. Most of us have heard of relative humidity and dewpoint temperature, but there are many other ways to measure the presence of water. This also applies to water, including water in gaseous form, which is the source of all life on our planet. It is said that a beloved child has many names. The following article explains the behavior of water vapor in air and clarifies the terminology used to describe it. It can also be measured in many ways and described with various terms. Water is known by different names in different states. Relative Humidity, Dewpoint, Mixing Ratio…. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
